 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
Metformin is an oral antihyperglycemic agent widely prescribed for the management of type 2 diabetes mellitus, functioning primarily by reducing hepatic glucose production and improving insulin sensitivity. Unlike insulin secretagogues, Metformin does not stimulate insulin release and thus carries a low risk of hypoglycemia. Ensuring the pharmaceutical purity of Metformin is essential to maintain its therapeutic effectiveness, minimize adverse effects, and meet regulatory standards. Impurity profiling plays a crucial role in detecting and controlling degradation products and synthesis-related by-products. Here are some of its known impurities listed below.
Showing 1 - 3 of 3 products
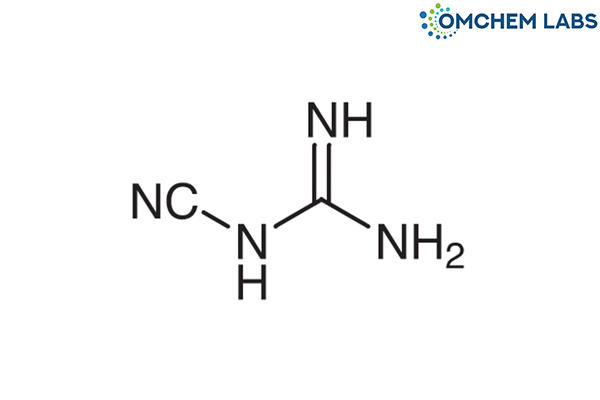
Catalogue No : METF-OCL-001
CAS No : 461-58-5
In Stock
Synonyms
Metformin EP Impurity A Metformin USP RC A 1-Cyanoguanidine
Metformin EP Impurity A Metformin USP RC A 1-Cyanoguanidine
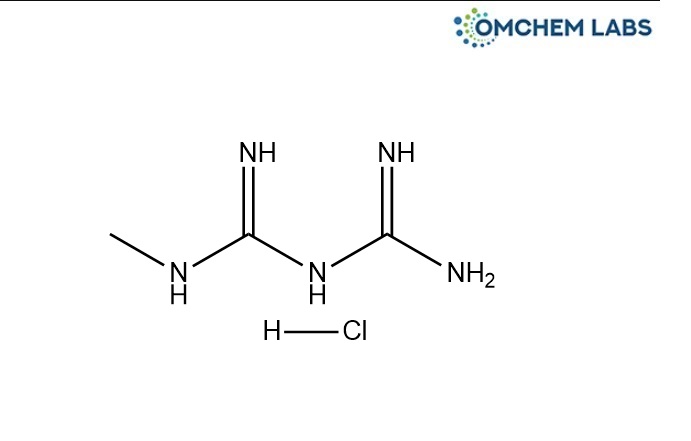
Catalogue No : METF-OCL-002
CAS No : 1674-62-0
In Stock
Synonyms
N-Desmethyl Metformin HCl 1-Methyl Biguanide HCl
N-Desmethyl Metformin HCl 1-Methyl Biguanide HCl
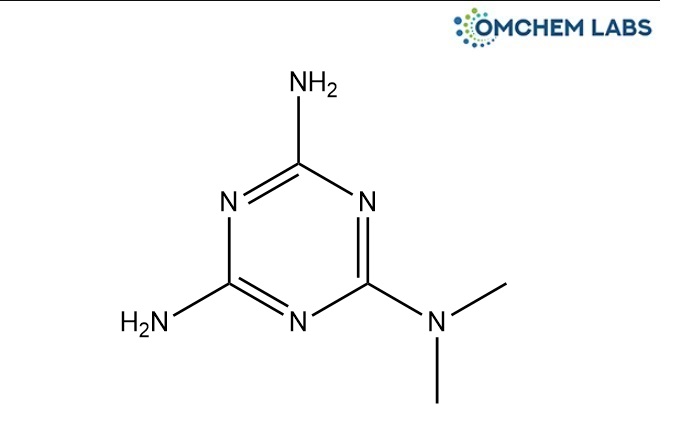
Catalogue No : METF-OCL-003
CAS No : 1985-46-2
In Stock
Synonyms
N,N-Dimethyl-1,3,5-triazine-2,4,6-triamine
N,N-Dimethyl-1,3,5-triazine-2,4,6-triamine
