 For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
For Laboratory Use Only: Intended strictly for research & development, and quality control purposes by qualified personnel only. Not for human or animal consumption, or for any medical, therapeutic, or diagnostic use.
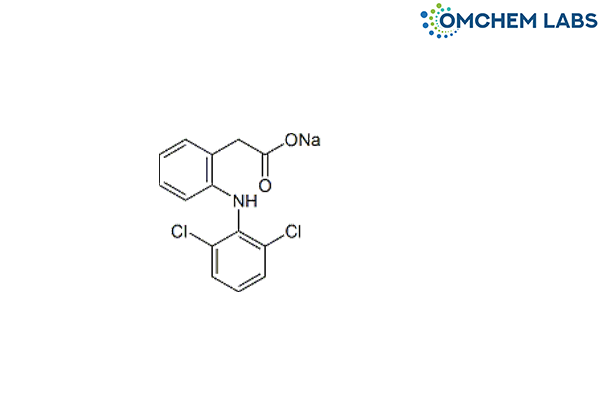
Aceclofenac Impurity A
| Catalogue No |
ACEC-OCL-002 |
| CAS NO |
15307-79-6 |
| Molecular Formula | C14H10Cl2NNaO2 |
| Molecular weight | 318.13 |
| Inquiry Status | In Stock |
| Synonyms | [2-[(2,6-Dichlorophenyl)amino]phenyl]acetic acid sodium salt |
Detailed Overview of this Impurity: Discover more about Impurity Standard & Analysis
Analytical Significance and Characterization of Aceclofenac Impurity A (CAS No. 15307-79-6) in Pharmaceutical Quality Control
Aceclofenac Impurity A, identified by CAS No. 15307-79-6, is a pharmacopoeial impurity formed during the synthesis or degradation of Aceclofenac, a widely used non-steroidal anti-inflammatory drug (NSAID). The presence and accurate quantification of such impurities are crucial for ensuring the safety, efficacy, and regulatory compliance of pharmaceutical formulations. This article presents a comprehensive overview of the origin, structure, analytical methods, and regulatory implications of Aceclofenac Impurity A, emphasizing its role in pharmaceutical quality control and drug development processes.
1. Introduction
Impurities in active pharmaceutical ingredients (APIs) can affect the safety and performance of drug products. Aceclofenac, a phenylacetic acid derivative, is used extensively for the management of pain and inflammation. During its synthesis, related substances such as Impurity A may form. According to ICH guidelines (Q3A and Q3B), the identification, qualification, and quantification of such impurities are mandatory. Understanding the nature and behavior of Aceclofenac Impurity A is essential for method development and validation in pharmaceutical analysis.
Aceclofenac Impurity A is structurally similar to Aceclofenac, differing primarily in functional groups or stereochemistry. The similarity necessitates precise analytical methods for separation and quantification.
3. Source and Formation
Impurity A typically arises during:
-
Synthesis: As a by-product or intermediate.
-
Degradation: Under thermal, photolytic, or oxidative stress conditions.
Process optimization and raw material control are essential to minimize its formation. Forced degradation studies help identify this impurity and understand its pathways.
4. Analytical Methods for Detection and Quantification
Several advanced analytical techniques are employed to detect and quantify Aceclofenac Impurity A:
-
High-Performance Liquid Chromatography (HPLC): The most widely used method, often with UV detection.
-
Liquid Chromatography-Mass Spectrometry (LC-MS/MS): For structural confirmation and trace-level detection.
-
NMR and IR Spectroscopy: To confirm the chemical structure.
Validation parameters include specificity, accuracy, linearity, limit of detection (LOD), and limit of quantification (LOQ), ensuring robust impurity profiling.
5. Regulatory Requirements
According to the ICH Q3A(R2) guideline:
-
Reporting thresholds typically start at 0.05% for APIs used at doses ≥2 g/day.
-
Qualification is required if the impurity exceeds specified thresholds.
-
Aceclofenac Impurity A may be listed in pharmacopeias like the European Pharmacopoeia or Indian Pharmacopoeia, with defined limits and reference standards.
6. Role in Pharmaceutical Quality Control
Monitoring Aceclofenac Impurity A ensures:
-
Product Safety: Preventing toxicological effects from unknown or unqualified impurities.
-
Batch Consistency: Ensuring reproducibility in manufacturing.
-
Regulatory Compliance: Meeting global standards for drug approval and marketing.
The use of certified reference standards for Aceclofenac Impurity A is critical for reliable analytical performance.
7. Conclusion
Aceclofenac Impurity A (CAS No. 15307-79-6) plays a vital role in the quality control of Aceclofenac formulations. Its identification, structural characterization, and quantification are fundamental to regulatory compliance and drug safety. As pharmaceutical guidelines continue to emphasize impurity profiling, advanced analytical techniques and standardization become increasingly important in ensuring the integrity of pharmaceutical products.
